Table of contents
- Introduction
- What is a pharmaceutical labeling workflow?
- Step 1 — Clinical trials and submission phase
- Step 2 — Advertising and promotional materials development
- Step 3 — Artwork and e-Labeling development
- Step 4 — Pre-press and printing phase
- Where most pharma labeling workflows break down
- How TVT fits into your pharma labeling ecosystem
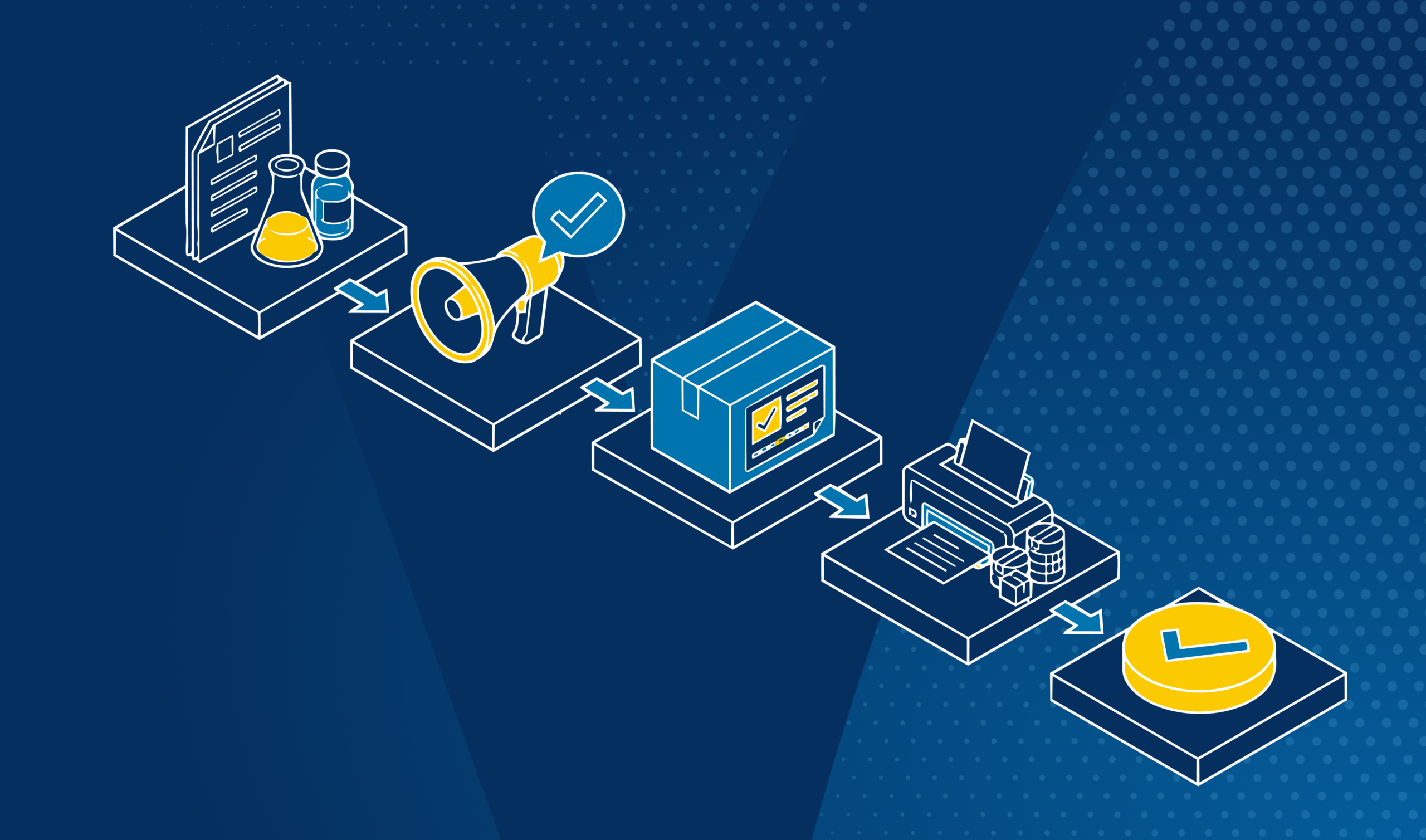
Pharmaceutical labeling is not a proofreading problem — it is a systems problem. With multiple stakeholders, regulatory jurisdictions, and high-stakes consequences for errors, understanding the full workflow is the first step to preventing failures.
Every year, labeling errors contribute to product recalls, regulatory rejections, and patient safety incidents across the pharmaceutical industry. The root cause is rarely a single missed typo — it is a fragmented workflow where regulatory content, artwork files, and print proofs travel through disconnected systems, reviewed by different teams with no unified verification layer.
This guide walks through the complete pharmaceutical labeling workflow, stage by stage, identifying where errors happen and how purpose-built verification technology like TVT can prevent them from reaching market.
What is a pharmaceutical labeling workflow? |
|---|
| A pharmaceutical labeling workflow is the end-to-end process by which drug labeling — from regulatory submissions to printed packaging — is created, reviewed, approved, and verified for compliance. It spans four core phases: |
| 1. Clinical submission — SmPC, PIL, and IFU documents prepared for health authorities |
| 2. Promotional and advertising materials — claims aligned to approved labeling |
| 3. Artwork and e-labeling — packaging design, multilingual translation, digital labeling |
| 4. Pre-press and print — final proofs verified before production |
Step 1 — Clinical trials and submission phase
The labeling lifecycle begins during clinical development. At this stage, companies prepare core regulatory documents — Summary of Product Characteristics (SmPC), Package Insert Leaflet (PIL), Instructions for Use (IFU), and clinical trial labeling — and submit them to health authorities such as the FDA, EMA, and national competent authorities.
This phase is characterized by frequent revisions. Safety language evolves as trial data accumulates, and the same information must be consistently reflected across multiple documents in multiple regulatory formats. A single inconsistency in safety wording between a dossier’s QRD template and its FHIR-formatted submission can trigger a query — or worse, a rejection.
Where errors happen in the submission phase
Where errors happen
- Misaligned documents across the dossier
- Missed updates when safety language changes
- Inconsistent wording across QRD, FHIR, SPL, SPM formats
How TVT supports this phase
- Many-to-one document comparison across submission files
- Automatic regulatory format recognition
- Full audit trail exportable as XML, PDF, or HTML
- Cross-submission consistency checks
Step 2 — Advertising and promotional materials development
Once a product approaches or achieves approval, marketing teams begin developing promotional materials — claims, Important Safety Information (ISI), branded communications, and regional adaptations. Every claim made in these materials must be traceable back to the approved label.
This is an area where labeling compliance frequently breaks down. Claims evolve through creative iterations, safety information gets reformatted, and accessibility requirements vary by market. Without systematic checking against the approved source, deviations accumulate undetected.
Where errors happen in promotional review
Where errors happen
- Claims drift from approved labeling text
- Missing or truncated safety information
- Formatting inconsistencies across regions
How TVT supports this phase
- Claim consistency checks against approved source text
- Deviations in embedded images detected automatically
- Font and number formatting validation
- Reading order verification for accessibility compliance
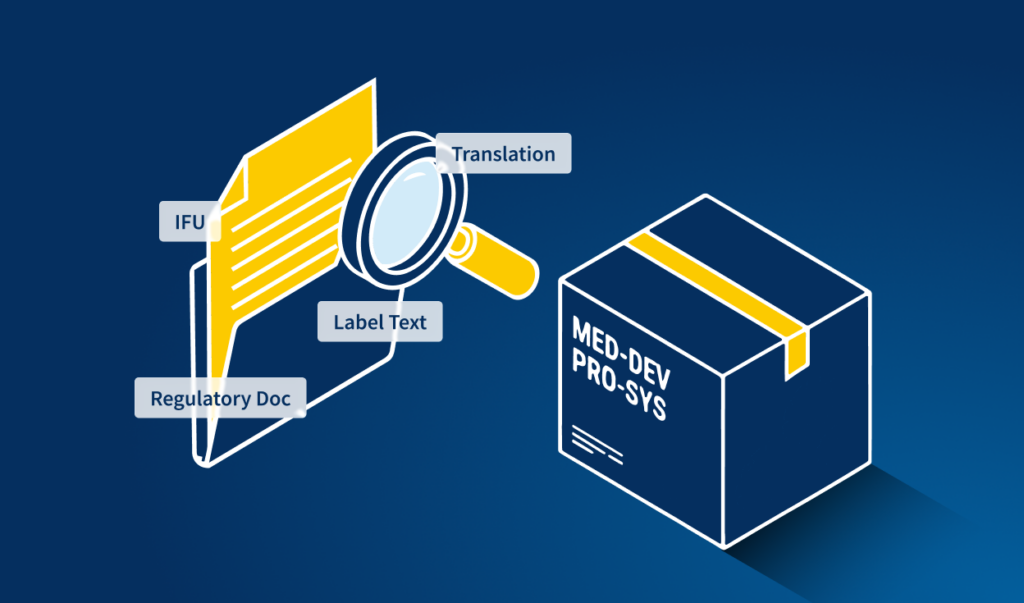
Step 3 — Artwork and e-Labeling development
The artwork phase is where regulatory text meets physical and digital packaging design. Cartons, labels, leaflets, and digital formats (ePI, eIFU) must accurately reproduce approved text — often translated into dozens of languages — while also meeting market-specific design and compliance requirements.
This is the highest-risk phase in the labeling workflow. Text is embedded in design files, translated by multiple vendors, and flows through artwork management systems that are rarely integrated with regulatory systems. Non-Latin scripts, DataMatrix barcodes, and multilingual leaflet layouts introduce failure points that visual inspection alone cannot reliably catch.
Where errors happen in artwork and e-labeling
Where errors happen
- Translation inconsistencies across language variants
- Non-Latin script rendering errors
- Missing or altered text in design files during typesetting
- Barcode URL errors (DataMatrix) and eIFU link failures
How TVT supports this phase
- One-to-many comparison of source regulatory text against all artwork files
- Multilingual proofreading across PDFs and all language versions
- Automated detection of text and artwork deviations
- DataMatrix barcode URL verification
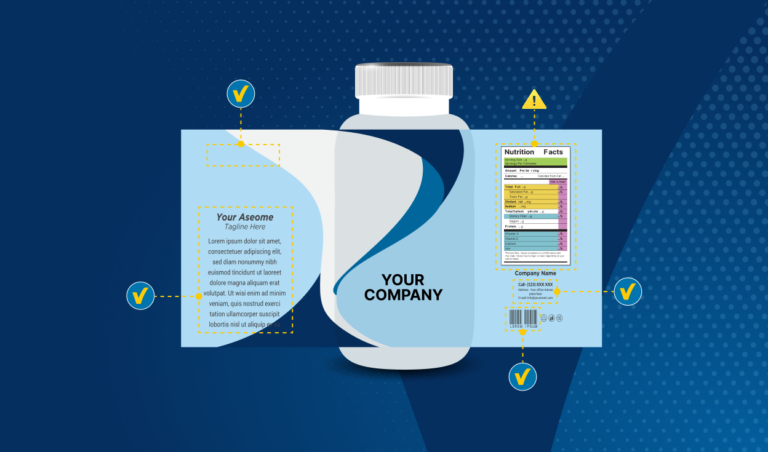
Step 4 — Pre-press and printing phase
At the pre-press stage, approved artwork files are handed off to contract printers and packaging suppliers. Printer proofs are generated and must be signed off before production runs begin. This handoff is a common source of final-minute errors — supplier-introduced changes, logo and color shifts, and layout adjustments that subtly alter the meaning of safety-critical content.
Without automated proof comparison, manufacturers must rely on visual inspection of complex multi-page documents, often under significant time pressure. An undocumented change at this stage can result in a batch recall or a regulatory non-conformance.
Where errors happen in pre-press and print
Where errors happen
- Supplier-introduced changes to approved artwork
- Logo and color deviations
- Layout shifts affecting meaning or readability of safety content
How TVT supports this phase
- Automated proof comparison against approved artwork
- Automated deviation annotations for rapid review
- Supplier compliance validation
- Full audit trail for batch documentation and regulatory inspection
Where most pharma labeling workflows break down
Labeling failures are rarely the result of negligence. They are the result of workflow architecture: systems that were not designed to talk to each other, teams working in parallel without shared visibility, and review processes that rely too heavily on human inspection of dense, complex documents.
- Manual proofreading limitations — Human reviewers cannot reliably detect all text deviations in multilingual documents, particularly in non-Latin scripts or numerically dense content.
- Disconnected regulatory and artwork systems — Regulatory content lives in one system; artwork lives in another. Changes in one rarely propagate automatically to the other.
- Lack of a unified audit trail — Without documented verification at each stage, reconstructing what was checked — and when — becomes difficult during audits or inspections.
- Late-stage error detection — Errors discovered at pre-press or post-batch are exponentially more costly to remediate than errors caught earlier in the workflow.
The following comparison illustrates the operational difference between manual review and a TVT-supported automated workflow across each phase:
Manual vs Automated Pharmaceutical Labeling Workflow |
||
|---|---|---|
| Stage | Manual Workflow | Automated Workflow (TVT) |
| Submission | Manual cross-checking of documents; high risk of inconsistency across regulatory formats | Automated multi-document comparison with regulatory format recognition |
| Claims review | Human-only validation; prone to drift across creative iterations | Automated consistency checks against approved source text |
| Artwork | Visual inspection of translated PDFs; non-Latin scripts difficult to review | Text and artwork verification across all languages and formats |
| Print proofs | Supplier trust; limited documentation of changes | Proof comparison with automated annotations and supplier compliance audit trail |
Navigating FDA guidelines for pharmaceutical packaging artwork
Learn how to navigate FDA guidelines for pharmaceutical packaging and artwork, from regulatory requirements to real-world risks, and how to ensure accuracy across every stage of the approval process.

How TVT fits into your pharma labeling ecosystem
TVT operates as a verification layer — not a system of record. It is designed to integrate alongside the platforms pharmaceutical companies already use, adding automated checking and audit documentation at each critical handoff point.
Works alongside
- Veeva Vault and document management platforms
- Artwork management systems
- Regulatory Information Management (RIM) systems
Position in the workflow
TVT sits between content creation and final approval — verifying that what was approved in one system is accurately represented in the next. It does not replace existing workflows; it strengthens them.
Key differentiators
- Many-to-one and one-to-many comparison modes
- Multilingual support including non-Latin scripts
- Regulatory format recognition across QRD, FHIR, SPL, and SPM
- Exportable audit trails in XML, PDF, and HTML formats
1. What is a pharmaceutical labeling workflow?
A pharmaceutical labeling workflow is the structured process by which drug labeling content is created, reviewed, translated, approved, and verified from initial regulatory submission through to printed or digital packaging. It spans multiple departments — Regulatory Affairs, QA, Artwork, and Marketing — and must comply with jurisdiction-specific requirements from health authorities such as the FDA, EMA, and national regulators. The workflow typically covers four phases: submission documentation, promotional materials, artwork and e-labeling, and pre-press print verification.
2. How do pharma companies ensure labeling compliance?
Pharmaceutical companies ensure labeling compliance through a combination of regulatory review, quality assurance sign-off, and automated verification technology. Compliance requires that all labeling content accurately reflects approved regulatory text, is consistently reproduced across all artwork and promotional formats, and is documented with a traceable audit trail. Standards such as GMP guidelines and regional regulatory requirements set the framework; automated tools like TVT provide the verification layer that ensures those standards are met at every stage of the workflow.
3. What causes errors in packaging and labeling?
Labeling errors most commonly arise from disconnected systems, manual review limitations, and late-stage error detection. When regulatory content is managed in one system and artwork in another, changes do not automatically propagate — leading to version mismatches. Manual proofreading of multilingual, design-embedded documents is unreliable, particularly for non-Latin scripts and numerically dense content. Supplier-introduced changes at pre-press, and missed updates when safety language evolves during the submission phase, are also frequent contributors to labeling failures.
4. How can automated proofreading reduce labeling errors?
Automated proofreading tools like TVT reduce labeling errors by systematically comparing source regulatory text against artwork, promotional materials, and print proofs — without relying on human visual inspection. They can detect text deviations in embedded PDFs, verify multilingual accuracy across dozens of language variants, validate barcode URLs, and produce documented deviation reports. Because automated comparison is consistent and exhaustive, it catches errors that human reviewers routinely miss. The result is earlier error detection, faster review cycles, and a defensible audit trail.
5. What systems are used in pharma artwork approval?
Pharmaceutical artwork approval typically involves a combination of content management, workflow, and verification systems. Veeva Vault is widely used as a document management and workflow platform for regulatory and artwork content. Artwork management systems manage the design and versioning of packaging files, while Regulatory Information Management (RIM) systems store approved labeling content. TVT operates as a dedicated verification layer across these systems — comparing approved regulatory text against artwork at each stage of the approval process and generating audit-ready reports of any deviations found.

FHIR-based ePI: A practical playbook for regulatory and labeling teams
A practical guide to FHIR-based ePI and how it’s reshaping pharmaceutical labeling and regulatory workflows.
Download the playbook