Table of contents
Errors in medical writing are not just editorial mistakes—they can lead to compliance issues, delayed approvals, and even patient safety risks.
From clinical documents and IFUs to regulatory submissions and training materials, medical writing must be precise, consistent, and aligned across multiple formats and versions.
That’s why proofreading in regulated environments goes far beyond spelling and grammar. It requires structured processes, version control, and the ability to detect even the smallest discrepancies.
This guide explains how to proofread medical writing effectively, and how regulated teams reduce risk while improving efficiency.
What is medical writing?
Medical writing refers to the creation of scientific, clinical, regulatory, and educational content used across the healthcare and life sciences industries.
It includes:
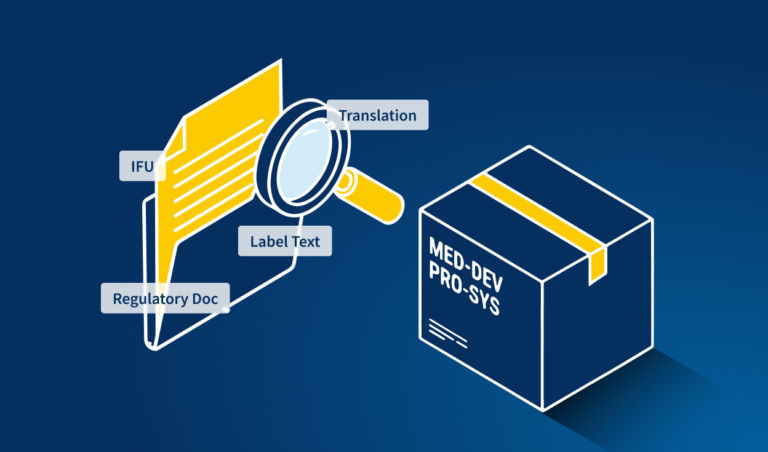
- Regulatory documents (e.g. clinical trial reports, submissions)
- Instructions for use (IFUs) and patient information leaflets
- Packaging and labeling content
- Educational and training materials
- Promotional and medical communications
Because this content directly impacts regulatory approval, product use, and patient safety, accuracy is critical at every stage.
Content verification software for medical device documentation
Get your medical device documentation, labeling, and packaging right the first time
Prepare complex content for global markets with fewer revisions and greater control.

Why proofreading medical writing is different
Proofreading medical writing is more complex than general proofreading because of the level of risk and precision involved.
Key challenges include:
- Technical terminology → medical and scientific language must be exact
- Numbers and units → dosage, strength, and measurements must be correct
- Regulatory wording → required phrases must appear exactly as approved
- Multilingual content → translations must remain aligned with source text
- Content reuse → the same text appears across multiple documents
- Formatting sensitivity → layout changes can alter meaning
Even small inconsistencies can lead to compliance issues, rework, or delays.
Medical writing target groups
Medical writing has very many target groups in both regulatory and non-regulatory contexts. Some of the key ones are:
- Healthcare Providers
- Patients
- Regulatory bodies
- Hospitals and other healthcare providers
- Insurance (health or otherwise) stakeholders
- Educational and training institutions
- Internal corporate stakeholders (e.g. within different functions of a pharmaceutical company or medical device manufacturer)
- Visitors to medical- and health-focused websites, or public events
- Industry press
Delving deeper into the different types of medical writing, most professionals agree on several categories that cover the whole range:
Regulatory and clinical documentation
This spans the various types of medical writing documents in research and development, clinical testing, submission, approval, and in the post-approval regulatory environment. Labeling and packaging, and instructions for use are very high-profile examples of this type of medical writing. It includes a hybrid of paper-based and digital (online) labeling.
Medical proofreading can be a very onerous task in this category, as inaccuracies can have drastic consequences for patients, and result in costly litigation for medical device manufacturers.
Educational and training materials
Examples are training manuals, slides, posters and other visual aids for various audiences, particularly training for Healthcare Providers. Video and multimedia scripts are other types in this category.
Medical communications
This includes documents such as press releases, white papers, case studies, health articles, marketing brochures, manuscripts, slide decks and posters, general health publications and general health websites. Research articles, reviews and case reports of individual patients are other types.
How to proofread medical writing (step-by-step)
1. Start from the approved source
Always use a single, validated source of truth for your content.
2. Define the reference version
Ensure everyone is working from the correct version to avoid confusion.
3. Compare versions systematically
Check updates against previous versions or approved master content.
4. Verify numbers, units, and symbols
Pay special attention to dosage, percentages, dates, and measurements.
5. Check formatting and structure
Review tables, headings, spacing, and layout-sensitive content.
6. Ensure consistency across documents
Verify that the same content is aligned across IFUs, labels, and related materials.
7. Document all changes
Maintain a clear record of edits, comments, and approvals.
8. Create an audit trail
Ensure the process is traceable and compliant with regulatory requirements.
A day in the life of a medical writer
Medical writing requires research, flexibility and often working to tough deadlines, especially in medical communications agencies, where multiple projects are the norm.
Joe Russo is a technical writer and information architect at Intuitive Surgical, best known for its da Vinci robotic surgical systems. He describes how a medical writing project might begin with a single 500-page document that he breaks down into single-page modules. These modules are topic-based and worked on by multiple medical writers simultaneously. This, he says, provides the agility the company needs in its process steps. He supports the medical writers with training, tools and output quality steps.
Typically, he will enter the process for producing product information at several points, initially by ensuring the medical writers have all medical device specifications for the document version. Finding synergies and overlaps is important. He looks at ways of re-using existing content from previous versions, as well as what information must be re-used across the different single-page modules. At the output stage, he supports medical writers through medical editing and updating style sheets for any changes that may have been made to a product after the medical writers have set to work. He also gives input on layout, tables and graphics.
Throughout the process, medical writers consult closely with the subject matter experts (SMEs). Product features, for instance, may change along the way. Typically, a project concludes with validation, verification and approval of the document modules. Tools used throughout these steps include standard software such as MS Office, but also Intuitive Surgical’s product lifecycle management system and TVT comparison software from Schlafender Hase® for proofreading and verifying documents. TVT makes the comparison of output incredibly easy, much simpler and much cleaner than other comparison tools, explains Joe Russo.
Tools and resources for medical writers
TVT, along with its modules TVT Artwork®, TVT Spelling® and TVT Barcode®, will compare any two document versions within seconds and highlight deviations in text, artwork and barcodes. Creating custom dictionaries is all possible in TVT, which is useful for the medical proofreading of terminology and corporate-specific nomenclature. It can be easily integrated into any information system, such as a product lifecycle management system. Annotation and report functions bring transparency to workflows.
In terms of online resources, in the American Medical Writers Association (AMWA) has a wealth of information, as does the European Medical Writers Association. Both are an excellent springboard for learning more about medical writing, medical proofreading and editing. Here, you can listen to Joe Russo, in full, talking about his job.
Would you like to find out more about how TVT comparison software can support you as a medical writer? Get in touch with us below.
What is proofreading in medical writing?
Proofreading in medical writing involves reviewing documents to ensure accuracy, consistency, and compliance with regulatory requirements.
Why is proofreading medical documents important?
Errors can lead to regulatory delays, product recalls, or patient safety risks, making accuracy critical.
What types of errors are hardest to detect manually?
Small changes in numbers, units, formatting, and repeated content across documents are often missed.
Can medical writing be proofread automatically?
Yes. Automated tools can compare documents, detect discrepancies, and support compliance workflows.
What is the difference between editing and proofreading?
Editing focuses on improving clarity and structure, while proofreading ensures accuracy and correctness before final approval.
References
1.https://info.amwa.org/ultimate-guide-to-becoming-a-medical-writer#what_is_medical_writing
See https://www.amwa.org for the American Medical Writers Association, and https://www.emwa.org for the European body.